When it comes to kidney disease, not everyone has the same risk. For people with recent African ancestry, there’s a hidden genetic factor that dramatically increases the chance of developing serious kidney problems - and it’s not about lifestyle, diet, or even high blood pressure alone. It’s called APOL1, and it’s one of the strongest genetic risk factors ever found for a common disease.
What Is APOL1 and Why Does It Matter?
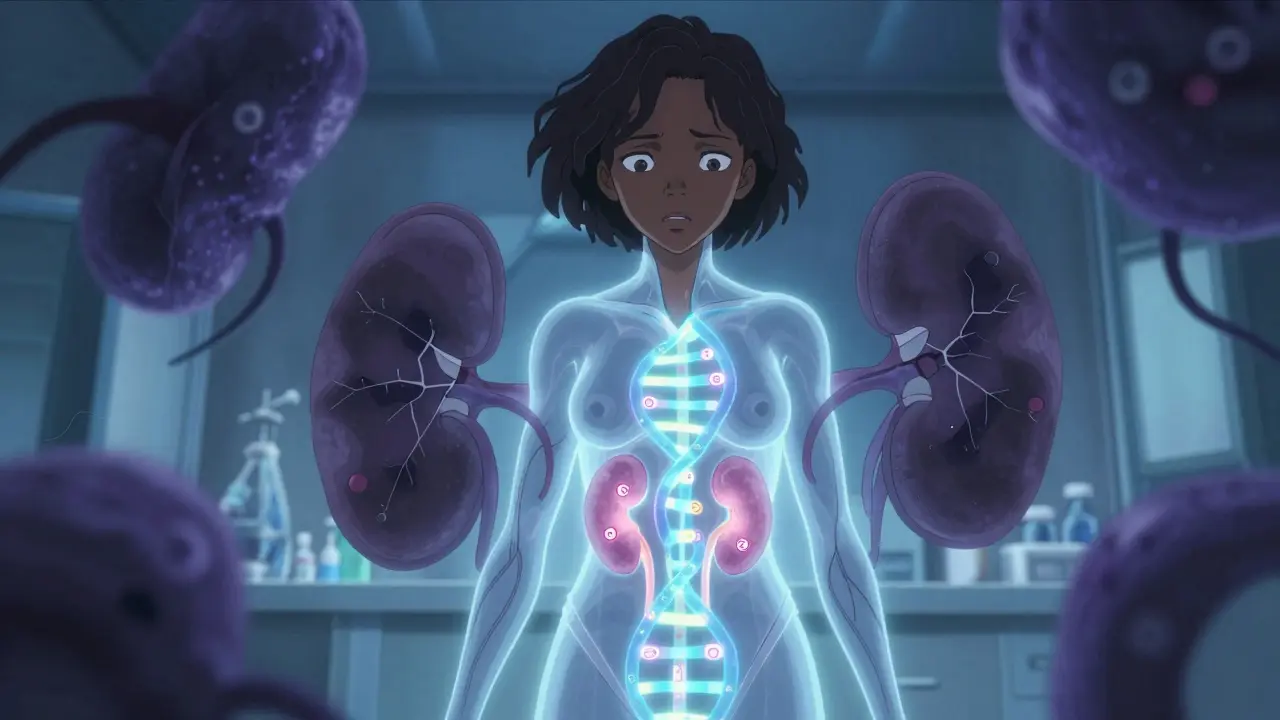
APOL1 is a gene that codes for a protein involved in your immune system. It helps fight off certain parasites, especially the one that causes African sleeping sickness. Around 3,000 to 10,000 years ago, two versions of this gene - called G1 and G2 - evolved in West and Central Africa. These versions gave people better protection against the parasite. But here’s the twist: the same changes that saved lives from infection now increase the risk of kidney damage.
Today, about 30% of people in Ghana and Nigeria carry these high-risk versions. In African Americans, about 13% have two copies of the risky variants (either G1/G1, G2/G2, or G1/G2). That might sound low, but here’s the kicker: among African Americans with non-diabetic kidney disease, nearly half have these high-risk genotypes. That means APOL1 explains about 70% of the extra kidney disease risk seen in people of African descent compared to others.
How Does APOL1 Cause Kidney Damage?
The APOL1 protein normally attacks parasites by punching holes in their membranes. But in people with the high-risk variants, the protein becomes overactive - and starts attacking kidney cells instead. This leads to scarring in the tiny filtering units of the kidneys, called glomeruli. The result? Conditions like focal segmental glomerulosclerosis (FSGS), collapsing glomerulopathy, and HIV-associated nephropathy (HIVAN).
What’s surprising is that most people with high-risk APOL1 genotypes never develop kidney disease. About 80% to 85% live with normal kidney function their whole lives. That means having the gene isn’t a death sentence - it’s a risk factor. Something else - like a viral infection (especially HIV), obesity, or high blood pressure - usually pushes the system over the edge. These are called "second hits." Without them, the gene alone often stays quiet.
Why Is This Risk Specific to African Ancestry?
These APOL1 variants are almost never found in people of European, Asian, or Indigenous American descent. That’s because they only evolved in populations exposed to the sleeping sickness parasite in sub-Saharan Africa. When enslaved Africans were forcibly brought to the Americas, they carried these genes with them. Today, the highest rates are seen in African Americans, Afro-Caribbeans, and other groups with recent West African roots.
It’s not race. It’s ancestry. And that distinction matters. Doctors used to assume kidney disease in Black patients was just due to high blood pressure or diabetes. But APOL1 research shows that even when those conditions are controlled, the genetic risk remains. This is why race-based kidney function estimates (like eGFR formulas that adjust for "Black race") are being phased out. The American Medical Association officially recommended dropping those adjustments in 2022 - because APOL1 gives us a better, more accurate way to understand risk.

Who Should Get Tested for APOL1?
Genetic testing for APOL1 became available in 2016. Today, it costs between $250 and $450 out-of-pocket. Insurance rarely covers it unless there’s a clear medical reason. So who should consider it?
- People of African ancestry with unexplained kidney disease (especially FSGS or collapsing glomerulopathy)
- Those with a family history of kidney failure
- Living kidney donors of African descent - testing is now recommended before donation to protect both donor and recipient
- People with HIV and kidney problems - nearly half of HIV-related kidney failure in African ancestry populations is tied to APOL1
But here’s the catch: getting tested isn’t always easy. A 2022 survey found that 78% of nephrologists felt unprepared to explain APOL1 results to patients. Many patients misunderstand their risk - thinking a positive test means they’ll definitely get kidney disease. In reality, only 15% to 20% of people with high-risk genotypes develop kidney failure in their lifetime. That uncertainty can be stressful.
What Can You Do If You Have High-Risk APOL1?
There’s no cure yet - but there are proven ways to reduce your risk.
- Control blood pressure. Keep it below 130/80 mmHg. Medications like ACE inhibitors or ARBs are often recommended.
- Monitor urine protein. Get an annual urine albumin-to-creatinine ratio (UACR) test. Rising protein levels are an early warning sign.
- Avoid kidney stressors. Don’t use NSAIDs (like ibuprofen) regularly. Avoid smoking. Manage weight and diabetes if you have them.
- Watch for infections. HIV and other viruses can act as "second hits." Stay up to date on vaccines and screenings.
One woman, Emani, found out she had high-risk APOL1 before any kidney damage occurred. She started regular check-ups, cut back on salt, and began taking blood pressure medication. Five years later, her kidney function is still normal. Early action made all the difference.

What’s Coming Next?
There’s real hope on the horizon. In October 2023, Vertex Pharmaceuticals announced promising results from a Phase 2 trial of a drug called VX-147. It’s the first treatment designed specifically to block the harmful effects of the APOL1 protein. In just 13 weeks, it reduced protein in the urine by 37% - a major sign that kidney damage may be slowing.
The NIH is now running a 10-year study called the APOL1 Observational Study (AOS), tracking 5,000 people with high-risk genotypes to better understand what triggers disease. By 2025, researchers aim to build tools that predict who’s most likely to progress - so interventions can be targeted. By 2027, the goal is to make testing accessible across all income levels, not just in wealthy countries.
Right now, only 12% of low- and middle-income countries offer APOL1 testing. That’s a huge gap. The science is here - but equity isn’t. If these new drugs become widely available, they could cut kidney failure rates in African ancestry populations by up to 35% by 2035. But only if everyone who needs them can get them.
Real Stories, Real Impact
One Reddit user, "BlackMedStudent," writes: "I’m a medical student with the high-risk genotype. I check my blood pressure every week. I get my urine tested every year. The knowledge scares me - but it also gives me power. I’m not waiting to find out if I’ll get sick. I’m doing something about it." Another user on the National Kidney Foundation forum said: "My doctor told me I had a 1 in 5 chance of kidney failure. That’s not a guarantee - but it’s enough to change my life." These aren’t just statistics. They’re real people living with uncertainty - and choosing to act.
Is APOL1 testing covered by insurance?
Most insurance plans do not routinely cover APOL1 testing unless there’s a clear clinical reason - like unexplained kidney disease or being considered as a living kidney donor. Out-of-pocket costs range from $250 to $450. Some labs offer payment plans or financial aid programs.
Can I pass APOL1 risk to my children?
Yes. APOL1 risk follows a recessive pattern. If both parents carry one risk variant, each child has a 25% chance of inheriting two copies (high-risk), a 50% chance of carrying one copy (low-risk carrier), and a 25% chance of having no risk variants. Genetic counseling is recommended before planning a family if you have a high-risk result.
Does having APOL1 risk mean I’ll definitely get kidney failure?
No. Only about 15% to 20% of people with two high-risk variants develop kidney disease in their lifetime. Most never do. The gene increases risk - it doesn’t guarantee disease. "Second hits" like infections, high blood pressure, or obesity are usually needed to trigger damage.
Why is APOL1 called an "unusual" disease gene?
It’s unusual because it evolved to protect against infection, not cause disease. It’s also ancestry-specific, has incomplete penetrance (most carriers don’t get sick), and follows a recessive pattern - unlike most inherited kidney disorders that are dominant and affect nearly everyone who carries the gene.
Are there any drugs to treat APOL1-related kidney disease?
Not yet approved - but promising. Vertex Pharmaceuticals’ drug VX-147 showed a 37% reduction in proteinuria in a 2023 trial, the first time a drug directly targeted the APOL1 protein. Phase 3 trials are underway. If successful, this could be the first treatment specifically for APOL1-mediated kidney disease.
Final Thoughts
APOL1 isn’t just another gene. It’s a window into how evolution shapes modern health. It shows us that what once saved lives - protection from a deadly parasite - can now put lives at risk in a different way. But knowledge changes outcomes. Testing, monitoring, and early action can delay or even prevent kidney failure. And with new drugs on the horizon, the future isn’t just about managing risk - it’s about stopping it before it starts.


Charity Hanson
February 28, 2026 AT 06:15As a Nigerian woman, I’ve always known kidney issues ran in my family-but never knew WHY until now. APOL1? That’s the missing piece. My aunt lost both kidneys by 42. My mom’s on meds at 58 and still kicking. This isn’t just science-it’s my story. If you’re African or of African descent, get tested. Not because you’re doomed, but because you deserve to outsmart your genes. I did. And I’m still here.
Noah Cline
February 28, 2026 AT 06:50Let’s be clear: APOL1 is a classic example of balancing selection with incomplete penetrance. The G1/G2 variants confer resistance to Trypanosoma brucei rhodesiense via pore-forming activity in the parasite’s lysosome. But in podocytes, the same mechanism induces mitochondrial dysfunction, ER stress, and autophagic collapse. The odds ratio for FSGS in homozygotes is 7.3 (95% CI: 5.1–10.4), per Genentech’s 2021 GWAS. We’re not talking about race-we’re talking about ancestral allele frequency in West African populations with >10% derived allele burden. Stop conflating ancestry with race. It’s epidemiologically lazy.
Lisa Fremder
February 28, 2026 AT 10:08Justin Ransburg
February 28, 2026 AT 12:50Thank you for this comprehensive overview. The distinction between ancestry and race is critical, and I appreciate how clearly it’s laid out. As a healthcare provider, I’ve seen too many patients mislabeled as having hypertensive nephrosclerosis when APOL1 was the real driver. Early detection through UACR screening and ACEi/ARB therapy can delay progression by over a decade. The upcoming VX-147 trials are a turning point-not just for treatment, but for equity in genomic medicine.
Sumit Mohan Saxena
March 1, 2026 AT 00:33From India, I find this deeply fascinating. While APOL1 variants are absent in South Asian populations, the broader principle of evolutionary trade-offs in immunity genes is universal. For instance, HLA-B27 in ankylosing spondylitis or G6PD deficiency in malaria-endemic regions follow similar patterns. The key insight here is not genetic determinism, but gene-environment interaction. The concept of 'second hits' aligns with the 'multiple hit hypothesis' in nephropathy pathogenesis. A well-structured piece, and a vital reminder that precision medicine must be ancestry-informed, not race-based.
Ajay Krishna
March 2, 2026 AT 16:27My uncle was a dialysis patient for 12 years. He never smoked, never had diabetes, ate clean. Then we found out-he had two copies of G1. That’s when I started pushing my family to get tested. Not because we’re scared, but because we’re smart. Knowledge isn’t fear. It’s power. And if you’re of African descent and haven’t checked your kidney health beyond a basic blood test? You’re playing Russian roulette with your kidneys. Get the UACR. Talk to your nephrologist. Don’t wait for the crash.
Brandon Vasquez
March 2, 2026 AT 22:59I’m a Black man in my late 30s. I got tested last year after my cousin went into kidney failure. Positive for G1/G2. I’ve been on lisinopril since. My BP is 122/78. My UACR is normal. I don’t talk about this much. But I show up. I monitor. I stay consistent. It’s not about fear. It’s about discipline. And yeah, I know most people with this won’t get sick. But I’m not betting on odds. I’m betting on myself.
Vikas Meshram
March 4, 2026 AT 06:28Actually, the APOL1 gene is not unique. Many genes have pleiotropic effects. For example, sickle cell trait protects against malaria but causes sickle cell disease in homozygotes. This is basic evolutionary biology. The fact that Western medicine only recently recognized this shows how biased medical research has been. The real issue is not the gene-it’s the decades of neglect in studying African populations. Until funding follows equity, we’ll keep having these delayed breakthroughs. Also, VX-147 is not a cure. It’s a bandaid. The real solution is universal healthcare access, not expensive gene-targeted drugs.
Ben Estella
March 6, 2026 AT 00:12bill cook
March 7, 2026 AT 13:59Charity Hanson
March 8, 2026 AT 20:34Hey @7897, I get it. I was terrified too. But here’s the thing: my sister got tested before pregnancy. She’s G1/G2. She’s on low-dose aspirin, BP meds, and sees a high-risk OB-GYN every 2 weeks. Her baby’s healthy. Her kidneys? Still perfect. It’s not about avoiding life. It’s about preparing for it. You can have kids. You just need a plan. And you’re not alone.
Byron Duvall
March 8, 2026 AT 23:59