RAS Blocker Combination Risk Assessment
Why This Matters
Combining ACE inhibitors and ARBs may seem logical for better blood pressure control, but research shows it increases serious risks without benefits. This tool helps assess your individual risk for adverse events like hyperkalemia or kidney damage.
The ONTARGET trial showed combining these drugs doubles hyperkalemia risk and increases acute kidney injury by 80%. There's no mortality benefit - only added danger.
Enter Your Current Health Information
Your Risk Assessment
When it comes to treating high blood pressure, heart failure, or kidney damage from diabetes, doctors often turn to two classes of drugs: ACE inhibitors and ARBs. Both target the same system in your body-the renin-angiotensin system (RAS)-but they do it in very different ways. And while they can seem interchangeable, mixing them together isn’t just a bad idea-it’s dangerous.
How ACE Inhibitors and ARBs Work
ACE inhibitors like lisinopril, enalapril, and ramipril block the enzyme that turns angiotensin I into angiotensin II. Angiotensin II is a powerful chemical that tightens blood vessels and tells your kidneys to hold onto salt and water. By stopping its production, these drugs lower blood pressure and reduce strain on the heart and kidneys.
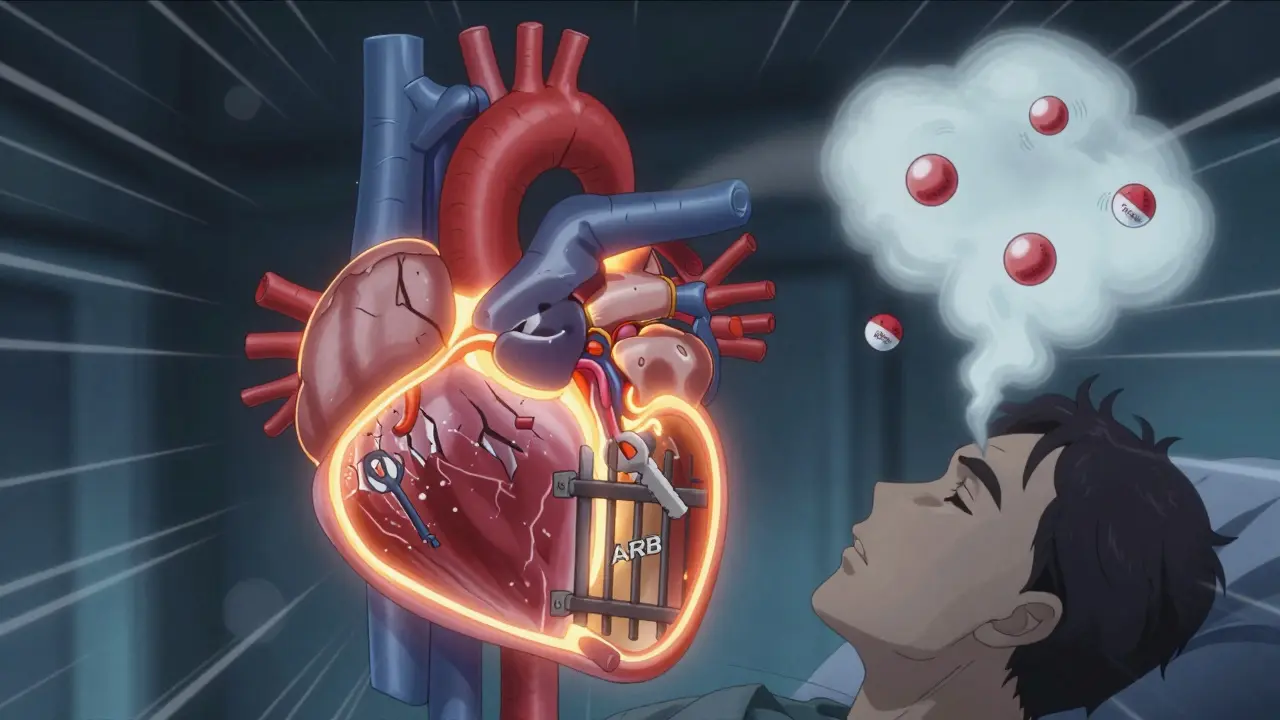
ARBs-like losartan, valsartan, and irbesartan-take a different approach. Instead of stopping angiotensin II from being made, they block its receptors. Think of it like this: ACE inhibitors stop the key from being made; ARBs lock the lock so the key can’t turn even if it’s there.
That small difference has big consequences. ACE inhibitors cause a buildup of bradykinin, a substance that can trigger a persistent dry cough in 10-15% of users. ARBs don’t do this, which is why many patients switch from an ACE inhibitor to an ARB when the cough becomes unbearable. The cough usually goes away within days of switching.
Side Effects That Set Them Apart
Both drugs can raise potassium levels and hurt kidney function, especially in people with existing kidney disease or diabetes. But their side effect profiles aren’t the same.
- ACE inhibitors: Dry cough (10-15%), angioedema (0.1-0.7%), higher risk of low blood pressure
- ARBs: Cough (3-5%), angioedema (0.1-0.2%), slightly less risk of dizziness
According to a 2019 Cochrane Review of over 146,000 patients, ARBs were discontinued less often due to side effects. For every 55 people treated with an ARB instead of an ACE inhibitor, one person avoided having to stop their medicine because of a bad reaction.
For heart failure patients, ACE inhibitors have stronger evidence for saving lives. Studies show they cut death risk by about 23%, while ARBs lower it by 15%. That’s why guidelines still put ACE inhibitors first for heart failure with reduced ejection fraction.
The Big Problem: Combining Them
It might sound logical-take both drugs to block the system twice as hard. But that’s not how it works. In reality, combining ACE inhibitors and ARBs doesn’t give you better outcomes. It just gives you more danger.
The ONTARGET trial in 2008 was a turning point. Researchers gave high-risk patients either ramipril (an ACE inhibitor), telmisartan (an ARB), or both. The combo group had lower blood pressure and less protein in their urine. But they also had:
- Double the risk of hyperkalemia (high potassium)
- 1.8 times more acute kidney injury
- More than double the chance of needing dialysis (2.3% vs. 1.0%)
No one lived longer. No one had fewer heart attacks or strokes. The extra blood pressure drop didn’t matter.
Since then, every major guideline has warned against this combo. The American Heart Association, the American College of Cardiology, and the European Society of Cardiology all say: Don’t do it. The 2023 ACC/AHA hypertension guidelines specifically call dual RAS blockade "contraindicated outside of clinical trials."

When Might It Still Be Considered?
There’s one tiny exception. A few nephrologists might consider adding an ARB to an ACE inhibitor in rare, non-diabetic cases where a patient has proteinuria over 1 gram per day and hasn’t responded to maximum ACE inhibitor therapy. Even then, it’s not standard.
Dr. Srinivasan Beddhu from the University of Utah has noted that in these rare cases, proteinuria can drop by 40-60%. But every patient who gets this combo needs weekly blood tests for potassium and kidney function. It’s high-risk, high-monitoring, and not for the average patient.
In real-world practice, most doctors have stopped doing it. A 2023 survey of 317 primary care doctors found that 89% had discontinued the combo after the 2018 VA NEPHRON-D trial showed a 27% increase in serious side effects with no kidney benefit.
What to Do Instead
If your blood pressure isn’t controlled on one RAS blocker, don’t add the other. Here’s what works better:
- Add a diuretic like hydrochlorothiazide or chlorthalidone
- Use a mineralocorticoid receptor antagonist like spironolactone (12.5 mg daily) - this cuts proteinuria by 30-40% with fewer risks
- Switch from an ACE inhibitor to an ARB if you have a cough
- For heart failure, consider ARNIs (angiotensin receptor-neprilysin inhibitors) like sacubitril/valsartan, which are now first-line in many cases
Spironolactone, for example, works on a different part of the system. It blocks aldosterone, which helps reduce fluid retention and kidney scarring without the same hyperkalemia risk as combining ACE inhibitors and ARBs.

Monitoring Is Non-Negotiable
Whether you’re on an ACE inhibitor or an ARB alone, you need regular blood tests. Check serum potassium and creatinine 1-2 weeks after starting or changing the dose. Then every 3 months if you’re stable.
High potassium (above 5.5 mmol/L) can cause dangerous heart rhythms. A sudden drop in kidney function (eGFR falling more than 30%) can mean your kidneys are struggling. Both are red flags.
One nephrologist at Massachusetts General Hospital reported discontinuing combo therapy in 87% of her 215 diabetic kidney disease patients because of high potassium or kidney decline. That’s not rare-it’s routine.
Market Trends and Future Directions
ACE inhibitors are still prescribed more often-58% of RAS blocker prescriptions in 2023 went to drugs like lisinopril. ARBs like losartan make up 42%. But the trend is shifting toward safer alternatives.
ARNIs (like Entresto) are replacing both in heart failure treatment. They’ve shown better survival rates than ACE inhibitors alone. New drugs targeting other parts of the RAS are in development, and by 2028, experts predict ACE inhibitor-ARB combinations will be used in less than 1% of cases.
The FDA has also flagged ARBs in the past for contamination with nitrosamines, which are cancer-causing impurities. While most of those recalls have been resolved, it’s another reminder: even small changes in manufacturing can affect safety.
Bottom Line
ACE inhibitors and ARBs are powerful tools. But they’re not interchangeable, and they’re definitely not additive. Using them together doesn’t make you healthier-it makes you sicker.
If you’re on one and it’s working, stick with it. If you have a cough, switch to an ARB. If your blood pressure isn’t controlled, add a diuretic or spironolactone-not another RAS blocker. And if someone suggests combining them, ask why. The evidence is clear: the risks outweigh any tiny benefit.
For most people, the best path isn’t doubling down-it’s choosing the right single agent and managing it well.


APRIL HARRINGTON
March 10, 2026 AT 13:34Like seriously I thought I had lung cancer
Switched to losartan and it vanished overnight
Why do doctors even still push ACE inhibitors if they know this happens?
Leon Hallal
March 11, 2026 AT 17:28Peter Kovac
March 11, 2026 AT 21:43Judith Manzano
March 13, 2026 AT 15:21Morgan Dodgen
March 14, 2026 AT 07:04Philip Mattawashish
March 15, 2026 AT 10:54Tom Sanders
March 16, 2026 AT 15:47Jazminn Jones
March 17, 2026 AT 00:51Stephen Rudd
March 17, 2026 AT 15:57Erica Santos
March 19, 2026 AT 09:08George Vou
March 19, 2026 AT 13:04Scott Easterling
March 19, 2026 AT 22:07Mantooth Lehto
March 20, 2026 AT 01:23Melba Miller
March 20, 2026 AT 08:46Katy Shamitz
March 22, 2026 AT 00:26